As a nasty fight over Planned Parenthood rages in Congress, the "abortion pill" that was once supposed to end the debate celebrates its 15th anniversary, embattled as ever.
RU486 supporters promised "abortion would become simple and easy," Beverly Winikoff of Gynuity Health Projects wrote in the September issue of the journal Contraception. Instead of trips to clinics surrounded by picketers, it was thought, women would just pick up a prescription. Critics, meanwhile, feared that women would "run rampant, having more abortions than ever," Winikoff added. Boyfriends would slip the stuff into their girlfriends' tea, and worse.
None of that happened. "It turns out women don't say, 'Hey, now that there's a pill for it, I think I'll have an abortion,'" Winikoff told BuzzFeed News.
The availability of the abortion pill has not led to a surge in U.S. abortions, which numbered about 1.3 million in 2000, when the pill was introduced, and 1.1 million in 2011. But the FDA's approval of RU486, or mifepristone, did spur a quiet revolution in how abortion is practiced. These pills, which now account for about one-third of early abortions in the U.S., have helped shift abortion to earlier stages of pregnancy.
Almost 90% of U.S. abortions are first-trimester procedures, either done with pills (also known as a "medical abortion") or a 15-minute surgical procedure, performed under local anesthetic, that removes the fetus and placenta with suction.
In federal political battles, this reality of abortion practice has been largely overlooked. A congressional hearing on Thursday afternoon will debate two bills aimed at defunding Planned Parenthood and restricting the rarest of late-term abortions, the 1.2% of procedures done after 20 weeks of pregnancy.
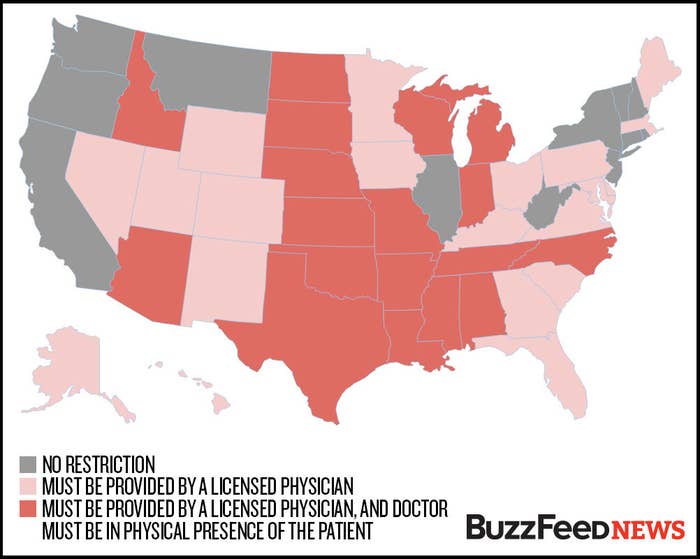
But in statehouses, the abortion pill has taken center stage. In 38 states, lawmakers have passed laws restricting medical abortions in various ways, mostly by requiring them to be facilitated by a medical doctor as opposed to a nurse. Several states have also mandated high and outdated dosages of the pills, and others have forced doctors to tell women, despite the lack of scientific backing, that a certain hormone can "reverse" the abortion procedure.
A medical abortion works in two steps.
According to a protocol outlined by the World Health Organization and the American College of Obstetrics and Gynecology, women in their first 63 days of pregnancy should first take a 200-milligram dose of mifepristone, followed 36 to 72 hours later by a second drug, misoprostol, which leads to bleeding and the end of the pregnancy.
In most states, a woman can take the second pill at home after a doctor's office visit. Medical abortions cost about $400, compared with $550 for early surgical abortions, and carry the same 0.1% rate of rare side effects such as infection.
"It's safer than penicillin," Winikoff said. "Insulin is more dangerous. Viagra is more dangerous."
Nevertheless, medical abortion faces restrictions nationwide, ones that trace to the curious conditions of RU486's FDA approval 15 years ago.
Like a hot potato, in 1994 the French drug maker Roussel-Uclaf handed off the rights to sell mifepristone in the U.S. to the Population Council, a nonprofit reproductive health group based in New York, in exchange for immunity from future lawsuits. The Population Council, in turn, licensed the drug to New York-based Danco Laboratories, which, after a battle with Congress, won approval of the drug in the waning days of the Clinton administration, and only with "black box" warning restrictions.
Today, the drug is available only from doctors trained in surgical abortion or blood transfusions, and it cannot be bought from pharmacies. In 38 states, laws require licensed medical doctors to provide the drug, despite the fact that the FDA's approval had allowed supervised nurses to do so.
In 18 states, the doctor must be in the physical presence of the woman while providing the pills, which prevents administration by televised medicine.
"All of this is really tied to anti-abortion feelings, and the potential of this drug [to make] abortion more accessible," Daniel Grossman, an OB-GYN at the University of California, San Francisco, told BuzzFeed News.
Three states — North Dakota, Ohio, and Texas — have legislated an additional restriction: that mifepristone be given before the 50th day of pregnancy, and at the 600-milligram dosage that was originally recommended when the FDA approved the drug in 2000.
Many doctors are outraged at these laws because that dosage is three times as high as what's recommended today, leading to more side effects, such as heavier bleeding. The laws also require three doctor visits instead of one.
Supporters of these laws, such as Michael Gonidakis of the Ohio Right to Life, have pointed to the FDA's 15-year-old dosage regimen as the "gold standard" for medical care. "What does it say about these abortionists that they think they know better than the FDA — that's pretty frightening," Gonidakis told BuzzFeed News.
But George Washington University's Susan Wood, a former assistant FDA commissioner for women's health, countered that medical practice routinely outpaces drug labels and that "off-label" use of drugs is an everyday part of medicine.
Mifepristone has turned out to be safer drug than the label initially indicated, Wood said. And in cases like these, the FDA usually doesn't intervene in physician practice, she added. "For medical abortion, changes need to come, in my opinion, because we now have better safety information."
In 2011, after 1.5 million women had taken the drug, just seven deaths (from infection) had been reported to the FDA — a very low mortality rate. Those deaths were all tied to vaginal delivery of the drug, so doctors now give it orally, lowering the risk further.
The FDA doesn't change labels unless the manufacturer asks, an uncertain, million-dollar process that might require lengthy safety studies. A roundup of independent safety studies of mifepristone reported in Contraception suggests the revision is overdue, but the cost and uncertainty of the process pose a high bar, Winikoff said.
Danco, as the license holder of RU486, could make the label request.
"Danco has discussions with FDA on a number of different issues, including possible revisions to the label," Danco representative Lauren Gray told BuzzFeed News by email. She did not give any timetable, however, saying that "details are confidential."

The restrictions on medical abortions are just some of the 282 laws since 2010 to limit abortions, according to the Guttmacher Institute.
Some of the laws require longer waiting periods before a woman can have an abortion. Others prohibit certain second-trimester abortions, or require abortion clinic doctors to have costly hospital-admitting privileges.
The effect of these laws is significantly fewer abortions in these states, according to a Contraception report led by Daniel Grossman of the University of California, San Francisco. In Texas, for example, where 19 of its 41 abortion clinics closed two years ago, the in-state rate of abortions dropped 13%, the study found.
And after Texas law mandated the higher dosage, medical abortions dropped from 70% to 28% of all abortions in the state. "Second-trimester abortions increased slightly, which is not the thing anyone wants," Grossman told BuzzFeed News.
Earlier this year lawmakers in Arkansas and Arizona passed one of the more unusual laws, which requires doctors to tell women that they could "reverse" a medical abortion after the first dose of mifepristone by taking a hormone called progesterone. Progesterone is sometimes given to pregnant women at high risk of an early delivery. (Woman change their minds only in about 0.004% of medical abortions, according to data gathered by Danco, Grossman said.)
But there is scant evidence that medical abortions can be stopped with progesterone. The claim is based on a 2012 study of six women who took the hormone after having a medical abortion, finding that four were able to carry their pregnancies to term. The report — which was led by a doctor who opposes abortion, George Delgado of Culture of Life Family Health Care in San Diego — said that more rigorous clinical trials were needed before doctors recommend using the hormone in this way.
Progesterone is probably unnecessary: If a woman takes the first pill in the medical abortion procedure but not the second, the pregnancy is likely to continue at about the same rate as what Delgado found in his study.
All of this means that the Arizona and Arkansas laws are pushing women into an unproven medical procedure, Grossman said, which "turns unethical experimentation on pregnant women into legislative mandate."
In April, Delgado told ThinkProgress he had case reports from another 200 women who followed his regimen and hoped to publish those results this month. He did not respond to a request for comment from BuzzFeed News.
Overall, while efforts to limit medical abortion have grown, so have the number of clinics that offer only mifepristone, Wood said. "They don't do other kinds of procedures, and they try and make the experience less of a visit to a health clinic and more of a visit to the doctor's office."
About 22% of Planned Parenthood clinics, for example, offer only medical abortions. An abortion clinic called Carafem in Chevy Chase, Maryland, advertises 24-hour medical abortions with subway ads saying "Abortion. Yeah, we do that."
Dc metro ad: never seen this before. I approve
The ads have stoked strong reactions from abortion opponents — HotAir called the responses "a mixture of horror and dark humor." But Wood pointed to the ad as an example of growing efforts to destigmatize abortion and point out the still-unrealized potential of RU486.
"The really good question about this anniversary is why, if this is so good, why is it unknown to so many people?" Winikoff said. The simple answer is that the pill isn't advertised, she added, and so most women don't learn about the option until they step into a clinic.
Despite the restrictions, medical abortions have grown more common over the last decade, from only 6% of all U.S. abortions in 2001 to 23% by 2011 and likely higher today, she added. "In that sense, it has been a quiet success."
